
Where ln 2 (the natural log of 2) equals 0.693. The relationship can be derived from decay law by setting N = ½ N o. There is a relation between the half-life (t 1/2) and the decay constant λ. In calculations of radioactivity one of two parameters ( decay constant or half-life), which characterize the rate of decay, must be known. , where N (number of particles) is the total number of particles in the sample, A (total activity) is the number of decays per unit time of a radioactive sample, m is the mass of remaining radioactive material. (Number of nuclei) N = N.e -λt (Activity) A = A.e -λt (Mass) m = m.e -λt The radioactive decay law can be derived also for activity calculations or mass of radioactive material calculations:

No matter how long or short the half life is, after seven half lives have passed, there is less than 1 percent of the initial activity remaining. Radioactive material with a short half life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. Notice that short half lives go with large decay constants.
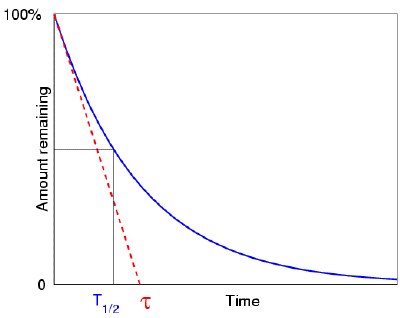
Half lives range from millionths of a second for highly radioactive fission products to billions of years for long-lived materials (such as naturally occurring uranium). In 14 more days, half of that remaining half will decay, and so on. If a radioisotope has a half-life of 14 days, half of its atoms will have decayed within 14 days. The half-life is the amount of time it takes for a given isotope to lose half of its radioactivity. The rate of nuclear decay is also measured in terms of half-lives. The radioactive decay of certain number of atoms (mass) is exponential in time. This constant probability may vary greatly between different types of nuclei, leading to the many different observed decay rates. This constant is called the decay constant and is denoted by λ, “lambda”. The radioactive decay law states that the probability per unit time that a nucleus will decay is a constant, independent of time. The fraction of the original activity remaining after succeeding half-lives is:Īctivity after 1 half-life = ½ of the originalĪctivity after 2 half-lives = ½ x ½ = ¼ of the originalĪctivity after 3 half-lives = ½ x ½ x ½ = (½) 3 = 1/8 of the originalĪctivity after 4 half-lives = (½) 4 = 1/16 of the originalĪctivity after 5 half-lives = (½) 5 = 1/32 of the originalĪctivity after 6 half-lives = (½) 6 = 1/64 of the originalĪctivity after 7 half-lives = (½) 7 = 1/128 of the original The number of atoms existing after 5 to 7 half-lives can usually be assumed to be negligible. After seven half-lives, only 1/128, or 0.78%, of the atoms remains. After five half-lives have elapsed, only 1/32, or 3.1%, of the original number of atoms remains.

Each radionuclide has its own particular half-life that never changes, regardless of the quantity or form of the material (i.e., solid, liquid, gas, element or compound) or its past history. Therefore, the rate of nuclear decay can be also measured in terms of half-lives. Thus, the probability of its breaking down does not increase with time, but stays constant no matter how long the nucleus has existed. A nucleus does not “age” with the passage of time. In other words, a nucleus of a radionuclide has no “memory”. As was written, radioactive decay is a random process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. The half-life is defined as the amount of time it takes for a given isotope to lose half of its radioactivity. One of the most useful terms for estimating how quickly a nuclide will decay is the radioactive half-life ( t 1/2).
